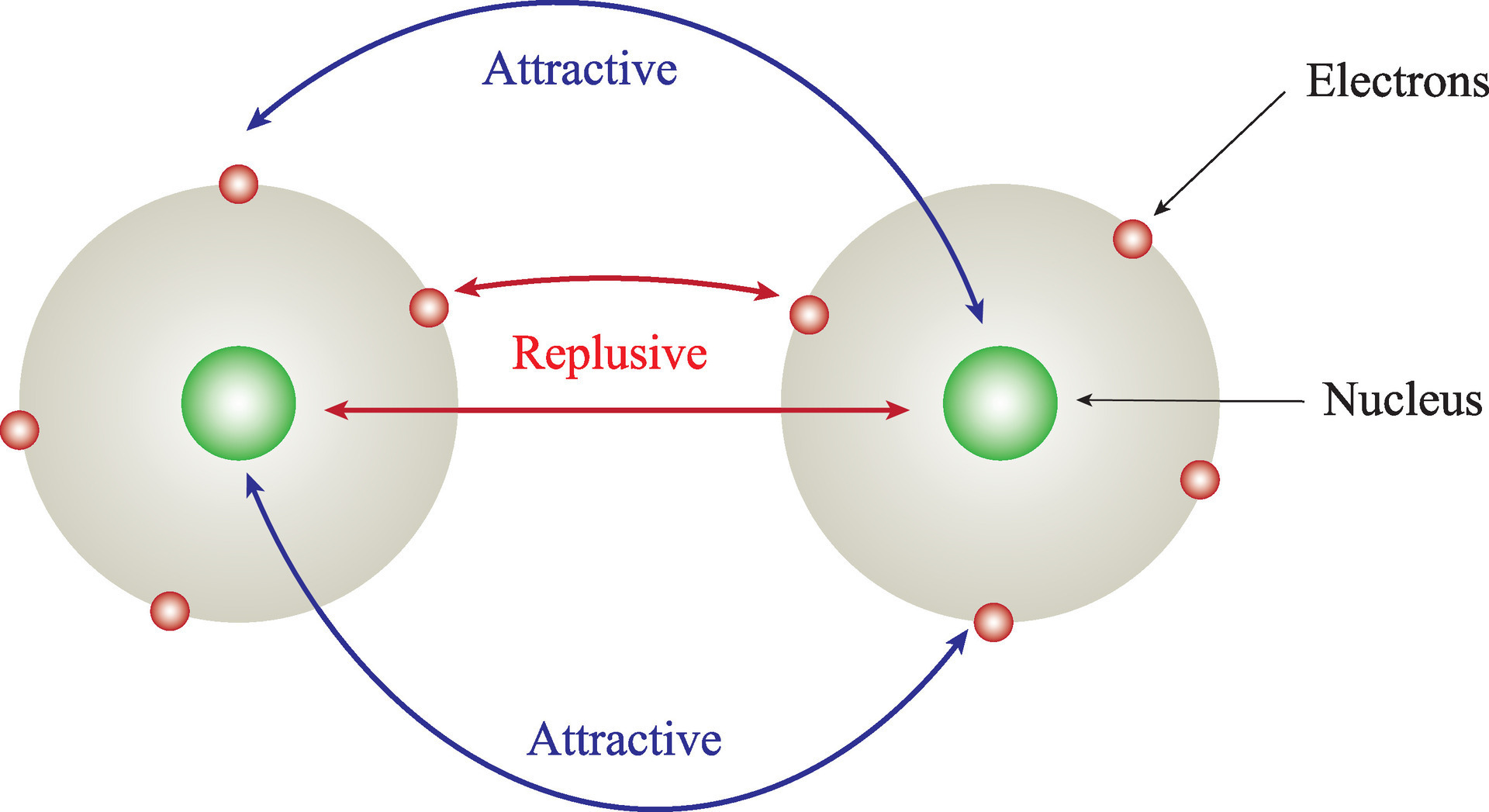
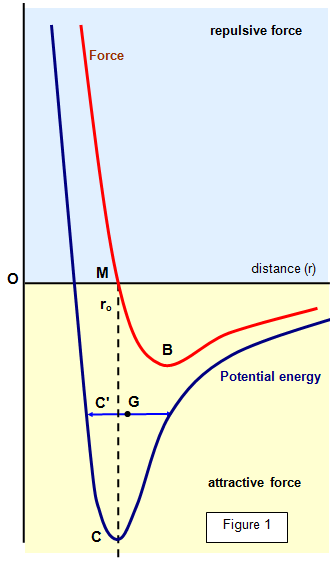
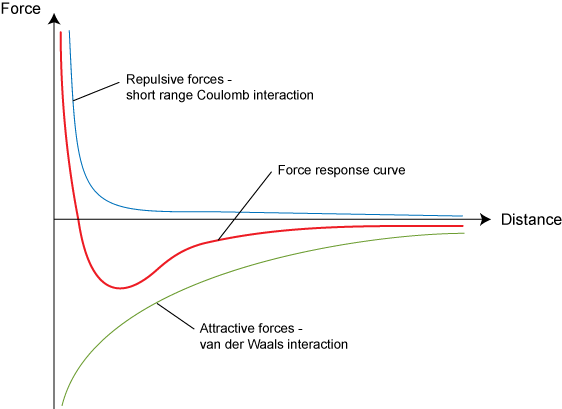
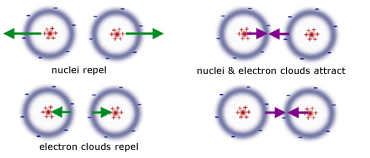
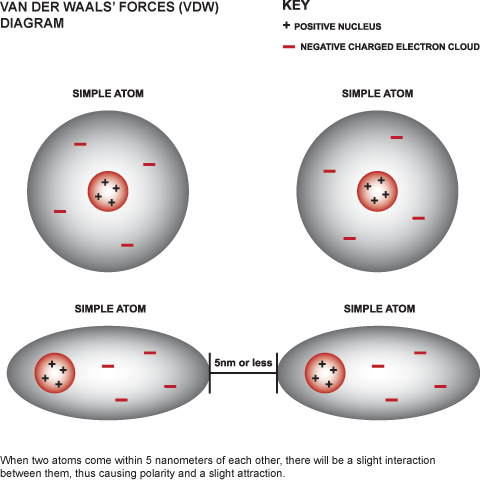
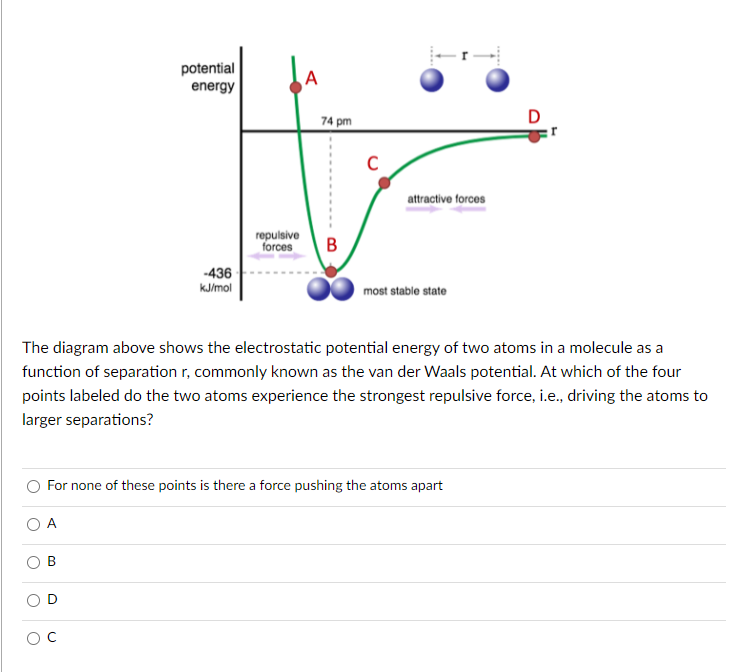
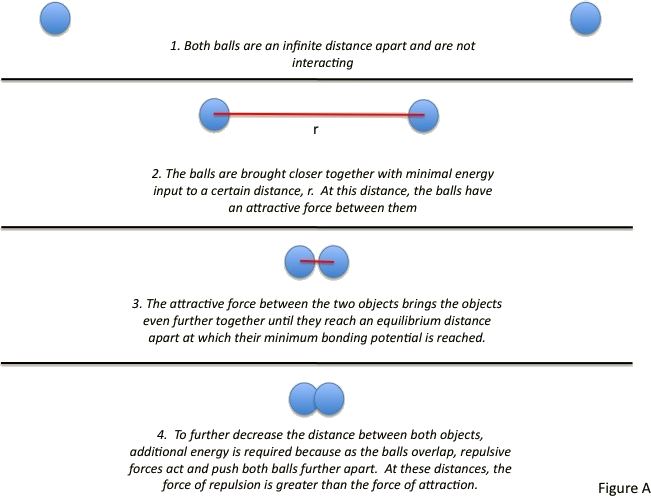
The combined repulsive and attractive van der Waals terms are described by plots of the potential energy as a function of interatomic separation distanceWhich of the following's assumptions can be drawn from

Magnetic Attraction Repulsion Force Law Magnets Stock Vector (Royalty Free) 2200459029 | Shutterstock
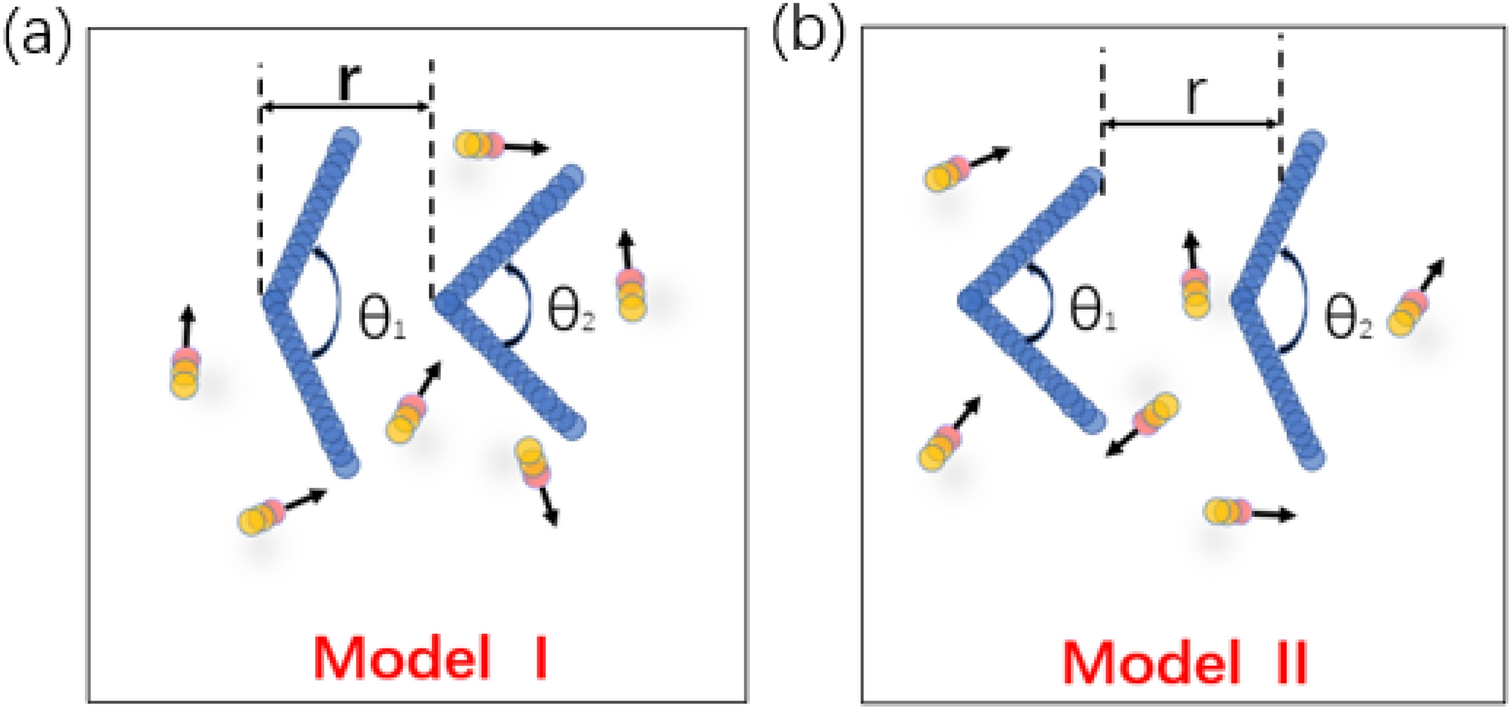
An attraction–repulsion transition of force on two asymmetric wedges induced by active particles | Scientific Reports
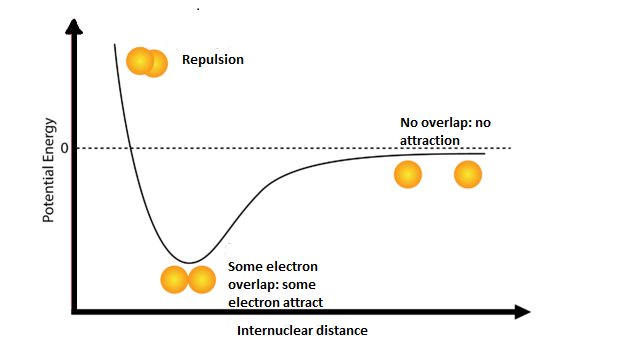
How do the attractive and repulsive forces between two atoms compare when the atoms form a covalent bond?

Attractive and repulsive forces between air bubble and mineral particle... | Download Scientific Diagram

What are the effects of the repulsive forces between electron on the potential energy graph for the dihydrogen molecule? - Chemistry Stack Exchange
What is the reason for the force of attraction or repulsion between two parallel wires carrying equal currents and placed near each other? - Quora